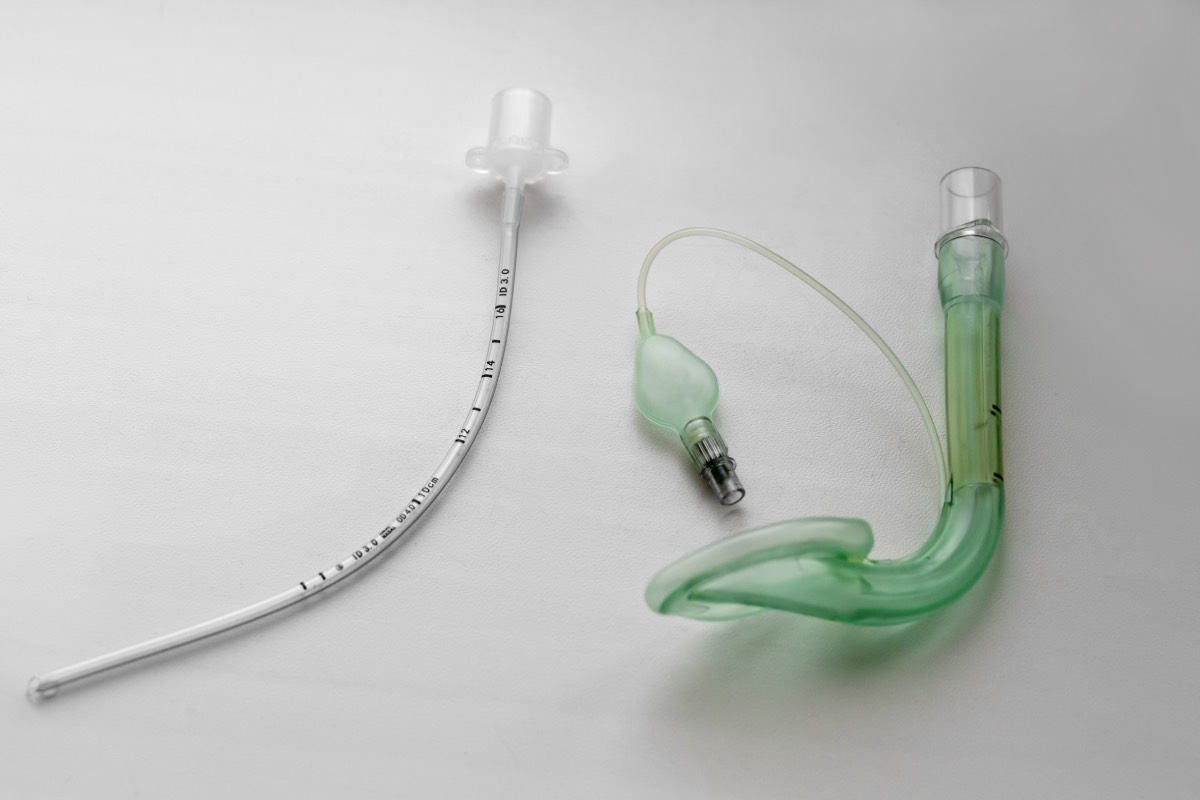
The endotracheal tube (ETT) and laryngeal mask airway (LMA) are two widely used devices for airway management during anesthesia and within emergency medicine. A review of medical literature reveals that while both the ETT and LMA are effective for maintaining airway patency, their clinical uses differ in some scenarios based on patient risk, procedural complexity, and desired outcomes.
The ETT is widely considered the gold standard for airway control, particularly in cases requiring complete airway protection and controlled ventilation. It is inserted through the vocal cords into the trachea, forming a sealed airway that minimizes the risk of aspiration. This makes it the preferred choice for procedures in which clinicians require full control over the patient’s airway. Literature consistently highlights its reliability in providing precise ventilation and oxygenation, especially during prolonged or complex surgical procedures.
In contrast, the LMA is a supraglottic airway device positioned above the vocal cords. Its design allows for easier and quicker placement without the need for laryngoscopy or muscle relaxants. As a result, LMAs are commonly used in elective surgeries, short-duration procedures, and situations where rapid airway access is needed. Studies show that LMAs can provide comparable ventilation to ETTs in some cases, particularly in low-risk patients undergoing routine procedures.
A key difference between ETT and LMA—and a significant driver of LMA use—is their complication profiles. Multiple randomized controlled trials and meta-analyses indicate that LMAs are associated with fewer perioperative complications, including reduced incidence of hypoxemia, postoperative cough, and hemodynamic instability. Additionally, LMAs tend to produce less airway irritation because they do not pass through the trachea, resulting in lower rates of sore throat and faster recovery times. These characteristics make LMAs particularly advantageous in outpatient and ambulatory surgery settings.
However, LMAs have important limitations. Because they do not provide a definitive seal within the trachea, they offer less protection against aspiration of gastric contents. This restricts their use in patients at risk of regurgitation, such as those with obesity, pregnancy, or gastrointestinal disorders. Furthermore, LMAs may be less effective in procedures requiring high airway pressures or in patients with poor lung compliance. In such cases, ETT remains the safer and more reliable option.
Clinical decision-making between ETT and LMA is therefore highly context dependent. For example, in pediatric and ambulatory surgeries, LMAs are often preferred due to ease of insertion, reduced stress response, and fewer postoperative complications. Conversely, in critical care, emergency airway management, or surgeries involving the airway itself, ETT is favored for its superior airway protection and ventilation control.
In summary, both ETT and LMA play essential roles in airway management. Their clinical uses overlap but are not equivalent, with ETT ensuring a secure airway that is indispensable in high-risk or complex clinical scenarios and LMA offering a less invasive alternative with fewer complications in appropriately selected patients. When choosing between these devices, the literature supports a tailored approach, emphasizing patient safety, procedural requirements, and clinician expertise.
References
- Zaman B, et al. Efficacy of laryngeal mask airway compared to endotracheal tube in airway management. Anesth Pain Med. 2022. https://doi.org/10.5812/aapm.120478
- Abid R, et al. Comparative study of airway management devices: ETT vs LMA. J Health Wellness Clin Res. 2025. https://doi.org/10.30476/beat.2024.102372.1509
- Dong W, et al. Comparison of laryngeal mask airway and endotracheal intubation during general anesthesia: A meta-analysis. Exp Ther Med. 2023. https://doi.org/10.3892/etm.2023.12253
- Zheng X, et al. Efficacy of laryngeal mask airway versus single-lumen tube in minimally invasive surgery. Sci Rep. 2025. https://doi.org/10.1038/s41598-025-10002-4
- Drake-Brockman TFE, et al. Laryngeal mask airway versus endotracheal tube in pediatric anesthesia. Lancet. 2017. https://doi.org/10.1016/s0140-6736(16)31719-6




